This is a post about our latest paper on marine microalgae
The value of Marine Microalgae
Marine microalgae are the pioneering photosynthetic organisms with significant morphological, genetic, and biochemical diversity. They are playing an important role in the biosphere, supplying nutrition to both aquatic and terrestrial food chains. Microalgae are ubiquitously distributed throughout the biosphere and exposed to high-oxygen and free-radical stresses, which has led to the evolution of efficient anti-oxidative defence mechanisms [1]. Marine algae living in the competitive harsh environments have developed specialised defence strategies by biosynthesising chemically and structurally diverse compounds through different metabolic pathways [2]. Microalgae are the “treasure house”, as rich sources of proteins, essential fatty acids, carbohydrates, pigments, vitamins, minerals, and several other bioactive molecules, with anti-oxidant, anti-inflammatory, antithrombotic, anti-bacterial, anti-diabetic, anti-fungal, anti-viral, anti-parasitic, anti-proliferative, anti-elastase, anti-trypsin, anti-chymotrypsin, angiotensin I-converting enzyme inhibitory (ACE-inhibitory), myofibroblast differentiation inducing, hepatic fibrosis inhibitory, etc. [3,4,5,6,7].
Microalgae and Inflammation
Our research hypothesis
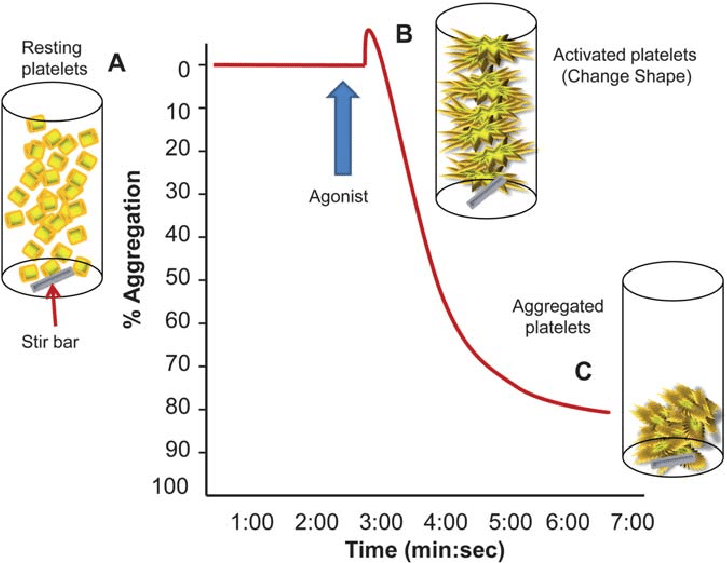
The marine Chlorococcum sp. lipids have not been tested yet towards the inflammatory and thrombotic pathways of PAF and thrombin. Therefore, in this manuscript, we have characterised the lipid fractions of marine microalga Chlorococcum sp. for anti-PAF and antithrombin activities with a view that the microalga can be an alternative sustainable source for the above bioactivities.
Conclusions
The in vitro assays of the present study showed that the glycolipid and phospholipid fractions of marine microalga Chlorococcum sp. SABC 012504 possess strong anti-PAF and antithrombin activities in human platelets. These bioactivities are likely due to the presence of specific novel Chlorococcum sp. SABC 012504 SQDG molecules, HexCer-t36:2 (t18:1/18:1 and 18:2/18:0) cerebrosides with a phytosphingosine base, phosphatidylcholine (PC), and phosphatidylethanolamine (PE) molecules in their lipid fractions. These bioactive PC and PE molecules with n-3 PUFA ALA or the MUFA OA were earlier reported from other natural sources, including marine, to possess strong antagonistic and agonistic effects against the PAF pathway of human platelet aggregation. Therefore, as evidenced in this study, the microalga Chlorococcum sp. has potential as a dietary supplement for these bioactivities.
References
- Becker, E.W. Microalgae: Biotechnology and Microbiology; Cambridge University Press: Cambridge, UK, 1994; ISBN 0-521-35020-4. [Google Scholar]
- Balandrin, M.F.; Klocke, J.A.; Wurtele, E.S.; Bollinger, W.H. Natural plant chemicals: Sources of industrial and medicinal materials. Science 1985, 228, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Antonopoulou, S.; Karantonis, H.C.; Nomikos, T.; Oikonomou, A.; Fragopoulou, E.; Pantazidou, A. Bioactive polar lipids from Chroococcidiopsis sp. (Cyanobacteria). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2005, 142, 269–282. [Google Scholar] [CrossRef] [PubMed]
- Koukouraki, P.; Tsoupras, A.; Sotiroudis, G.; Demopoulos, C.A.; Sotiroudis, T.G. Antithrombotic properties of Spirulina extracts against platelet-activating factor and thrombin. Food Biosci. 2020, 37, 100686. [Google Scholar] [CrossRef]
- Saha, S.K. Microalgae and cyanobacterial feedstocks for multiple commodities. In Feedstocks: Production Practices, Technologies and Environmental Impacts; Acquaye, A., Ed.; Nova: New York, NY, USA, 2014; pp. 73–107. [Google Scholar]
- Saha, S.K.; McHugh, E.; Murray, P.; Walsh, D.J. Microalgae as a source of nutraceuticals. In Phycotoxins: Chemistry and Biochemistry, 2nd ed.; Botana, L.M., Alfonso, A., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2015; pp. 255–292. [Google Scholar]
- Saha, S.K.; Murray, P. Exploitation of microalgae species for nutraceutical purposes: Cultivation aspects. Fermentation 2018, 4, 46. [Google Scholar] [CrossRef]
- Tsoupras, A.B.; Iatrou, C.; Frangia, C.; Demopoulos, C.A. The implication of platelet-activating factor in cancer growth and metastasis: Potent beneficial role of paf-inhibitors and antioxidants. Infect. Disord. Drug Targets 2009, 9, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.; Lordan, R.; Zabetakis, I. Inflammation, not Cholesterol, Is a Cause of Chronic Disease. Nutrients 2018, 10, 604. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Zabetakis, I. Thrombosis and COVID-19: The Potential Role of Nutrition. Front. Nutr. 2020, 7, 583080. [Google Scholar] [CrossRef]
- Zabetakis, I.; Lordan, R.; Norton, C.; Tsoupras, A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients 2020, 12, 1466. [Google Scholar] [CrossRef]
- Lordan, R.; Tsoupras, A.; Zabetakis, I. Platelet activation and prothrombotic mediators at the nexus of inflammation and atherosclerosis: Potential role of antiplatelet agents. Blood Rev. 2020, 100694, (online ahead of print). [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Zabetakis, I. Inflammation and Cardiovascular Diseases. In The Impact of Nutrition and Statins on Cardiovascular Diseases; Zabetakis, I., Lordan, R., Tsoupras, A., Eds.; Academic Press: Cambridge, MA, USA, 2019; Volume 3, pp. 53–117. [Google Scholar]
- Lordan, R.; Tsoupras, A.; Zabetakis, I. Phospholipids of animal and marine origin: Structure, function, and anti-inflammatory properties. Molecules 2017, 22, 1964. [Google Scholar] [CrossRef]
- Nasopoulou, C.; Tsoupras, A.B.; Karantonis, H.C.; Demopoulos, C.A.; Zabetakis, I. Fish polar lipids retard atherosclerosis in rabbits by down-regulating paf biosynthesis and up-regulating paf catabolism. Lipids Health Dis. 2011, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.P.; Chen, F.; Liu, X.; Lic, X.Z. Carotenoid composition in the green microalga Chlorococcum. Food Chem. 2002, 76, 319–325. [Google Scholar] [CrossRef]
- Zhang, D.H.; Lee, Y.K. Ketocarotenoid production by a mutant of Chlorococcum sp. in an outdoor tubular photobioreactor. Biotechnol. Lett. 1999, 21, 7–10. [Google Scholar] [CrossRef]
- Saha, S.K.; McHugh, E.; Hayes, J.; Moane, S.; Walsh, D.; Murray, P. Effect of various stress-regulatory factors on growth and lipids biosynthesis in microalga Haematococcus pluvialis. Bioresour. Technol. 2013, 128, 118–124. [Google Scholar] [CrossRef]
- Beevi, U.S.; Sukumaran, R.K. Cultivation of the fresh water microalga Chlorococcum sp. RAP13 in sea water for producing oil suitable for biodiesel. J. Appl. Phycol. 2015, 27, 141–147. [Google Scholar] [CrossRef]
- Antonopoulou, S.; Nomikos, T.; Oikonomou, A.; Kyriacou, A.; Andriotis, M.; Fragopoulou, E.; Pantazidou, A. Characterization of bioactive glycolipids from Scytonema julianum (cyanobacteria). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2005, 140, 219–231. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Shiels, K.; Saha, S.K.; Nasopoulou, C.; Zabetakis, I. In Vitro Antithrombotic Properties of Salmon (Salmo salar) Phospholipids in a Novel Food-Grade Extract. Mar. Drugs 2019, 17, 62. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Demuru, M.; Shiels, K.; Saha, S.K.; Nasopoulou, C.; Zabetakis, I. Structural Elucidation of Irish Organic Farmed Salmon (Salmo salar) Polar Lipids with Antithrombotic Activities. Mar. Drugs 2018, 16, 176. [Google Scholar] [CrossRef]
- Li, Y.; Hua, X.; Han, F.; Mu, J.; Di, C.; Bo, F.; Zeng, H. Regulation of lipid metabolism in the green microalga Chlorella protothecoides by heterotrophy–photoinduction cultivation regime. Bioresour. Technol. 2015, 192, 781–791. [Google Scholar] [CrossRef]
- Hu, C.-W.; Chuang, L.-T.; Yu, P.-C.; Chen, C.-N.N. Pigment production by a new thermotolerant microalga Coelastrella sp. F50. Food Chem. 2013, 138, 2071–2078. [Google Scholar] [CrossRef]
- Ra, C.H.; Kang, C.-H.; Kim, N.K.; Lee, C.-G.; Kim, S.-K. Cultivation of four microalgae for biomass and oil production using a two-stage culture strategy with salt stress. Renew. Energy 2015, 80, 117–122. [Google Scholar] [CrossRef]
- Mehariya, S.; Sharma, N.; Iovine, A.; Casella, P.; Marino, T.; Larocca, V.; Molino, A.; Musmarra, D. An Integrated Strategy for Nutraceuticals from Haematococcus pluvialis: From Cultivation to Extraction. Antioxidants 2020, 9, 825. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.O.; Han, J.G.; Kim, Y.; Ha, J.H.; Kim, S.S.; Jeong, M.H.; Jeong, H.S.; Kim, N.Y.; Cho, J.S.; Yoon, W.B.; et al. Lipid production in Porphyridium cruentum grown under different culture conditions. J. Biosci. Bioeng. 2009, 108, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Velea, S.; Ilie, L.; Filipescu, L. Optimization of Porphyridium purpureum culture growth using two variables experimental design: Light and sodium bicarbonate. UPB Sci. Bull. 2011, 73, 81–94. [Google Scholar]
- Huo, S.; Basheer, S.; Liu, F.; Elshobary, M.; Zhang, C.; Qian, J.; Xu, L.; Arslan, M.; Cui, F.; Zan, X.; et al. Bacterial intervention on the growth, nutrient removal and lipid production of filamentous oleaginous microalgae Tribonema sp. Algal Res. 2020, 52, 102088. [Google Scholar] [CrossRef]
- Tsoupras, A.; O’Keeffe, E.; Lordan, R.; Redfern, S.; Zabetakis, I. Bioprospecting for Antithrombotic Polar Lipids from Salmon, Herring, and Boarfish By-Products. Foods 2019, 8, 416. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.; Lordan, R.; O’Keefe, E.; Shiels, K.; Saha, S.K.; Zabetakis, I. Structural Elucidation of Irish Ale Bioactive Polar Lipids with Antithrombotic Properties. Biomolecules 2020, 10, 1075. [Google Scholar] [CrossRef] [PubMed]
- Vasange, M.; Rolfsen, W.; Bohlin, L. A sulphonoglycolipid from the fern Polypodium decumanum and its effect on the platelet activating-factor receptor in human neutrophils. J. Pharm. Pharmacol. 1997, 49, 562–566. [Google Scholar] [CrossRef]
- Bohlin, L.; Rolfsen, W.; Vasange, M. Use of Sulpho-Quinovosyl-Diacylglycerols for the Treatment of Inflammatory Skin Diseases. EP0952836B1, 28 October 2004. [Google Scholar]
- Berge, J.P.; Debiton, E.; Dumay, J.; Durand, P.; Barthomeuf, C. In vitro anti-inflammatory and anti-proliferative activity of sulfolipids from the red alga Porphyridium cruentum. J. Agric. Food Chem. 2002, 50, 6227–6232. [Google Scholar] [CrossRef]
- Oku, H.; Wongtangtintharn, S.; Iwasaki, H.; Shimatani, M.I.M.; Toda, T. Tumor specific cytotoxicity of glucosylceramide. Cancer Chemother. Pharmacol. 2007, 60, 767–775. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Harrington, J.; Pienaar, R.; Devaney, K.; Heaney, S.; Koidis, A.; Zabetakis, I. The Effects of Oxidation on the Antithrombotic Properties of Tea Lipids Against PAF, Thrombin, Collagen, and ADP. Foods 2020, 9, 385. [Google Scholar] [CrossRef] [PubMed]
- Rippka, R.; Stanier, R.Y.; Deruelles, J.; Herdman, M.; Waterbury, J.B. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloan-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissue. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Tsoupras, A.; Zabetakis, I.; Lordan, R. Platelet aggregometry assay for evaluating the effects of platelet agonists and antiplatelet compounds on platelet function in vitro. MethodsX 2018, 6, 63–70. [Google Scholar] [CrossRef] [PubMed]