1. Forty Years Since the Structural Elucidation of Platelet-Activating Factor (PAF): Historical, Current, and Future Research Perspectives
In the late 1960s, Barbaro and Zvaifler described a substance that caused antigen induced histamine release from rabbit platelets producing antibodies in passive cutaneous anaphylaxis. Henson described a ‘soluble factor’ released from leukocytes that induced vasoactive amine release in platelets. Later observations by Siraganuan and Osler observed the existence of a diluted substance that had the capacity to cause platelet activation. In 1972, the term platelet-activating factor (PAF) was coined by Benveniste, Henson, and Cochrane. The structure of PAF was later elucidated by Demopoulos, Pinckard, and Hanahan in 1979. These studies introduced the research world to PAF, which is now recognised as a potent phospholipid mediator. Since its introduction to the literature, research on PAF has grown due to interest in its vital cell signalling functions and more sinisterly its role as a pro-inflammatory molecule in several chronic diseases including cardiovascular disease and cancer. As it is forty years since the structural elucidation of PAF, the aim of this review is to provide a historical account of the discovery of PAF and to provide a general overview of current and future perspectives on PAF research in physiology and pathophysiology.
Our full paper is here.
2. Effect of acyl and alkyl analogs of platelet-activating factor (PAF) on inflammatory signaling
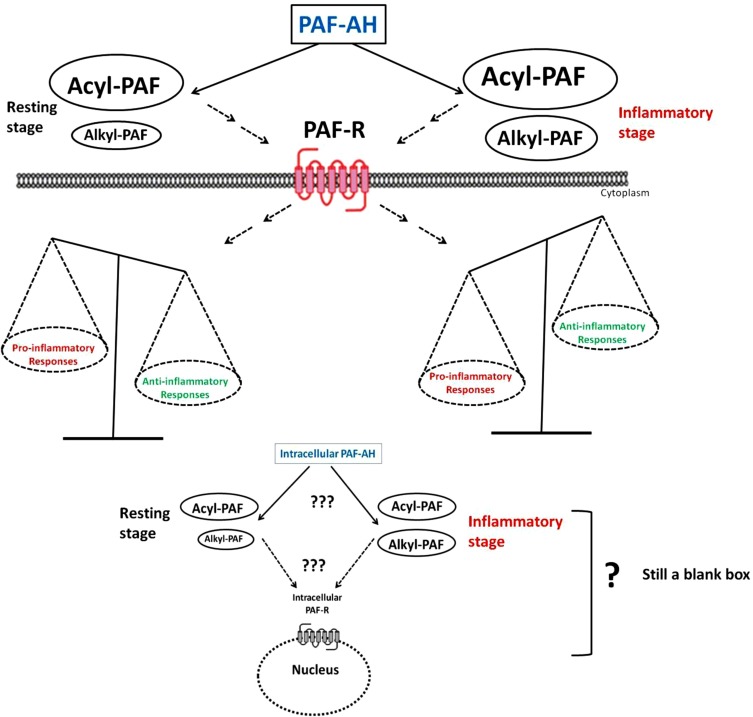
Platelet-activating factor (PAF), a bioactive ether phospholipid with significant pro-inflammatory properties, was identified almost half a century ago. Despite extensive study of this autocoid, therapeutic strategies for targeting its signaling components have not been successful, including the recent clinical trials with darapladib, a drug that targets plasma PAF-acetylhydrolase (PAF-AH). We recently provided experimental evidence that the previously unrecognized acyl analog of PAF, which is concomitantly produced along with PAF during biosynthesis, dampens PAF signaling by acting both as a sacrificial substrate for PAF-AH and probably as an endogenous PAF-receptor antagonist/partial agonist. If this is the scenario in vivo, PAF-AH needs to catalyze the selective hydrolysis of alkyl-PAF and not acyl-PAF. Accordingly, different approaches are needed for treating inflammatory diseases in which PAF signaling is implicated. The interplay between acyl-PAF, alkyl-PAF, PAF-AH, and PAF-R is complex, and the outcome of this interplay has not been previously appreciated. In this review, we discuss this interaction based on our recent findings. It is very likely that the relative abundance of acyl and alkyl-PAF and their interactions with PAF-R in the presence of their hydrolyzing enzyme PAF-AH may exert a modulatory effect on PAF signaling during inflammation.
The full paper is here.
3. Highlights on the role of PAF
- Despite its discovery more than 50 years ago, the role of PAF in inflammation is still not understood.
- Because PAF is a juxtacrine signaling molecule, correlating its levels to a disease state is challenging.
- Acyl-PAF, despite its unrecognized role in the past, can profoundly affect signaling through the PAF-R.
- It is clear from the wealth of evidence presented that understanding the mechanisms of PAF/PAF-R signalling in health and disease has yet to be fully elucidated. However, the discovery of various PAF and PAFLL agonists and antagonists, the role of the PAF metabolic enzymes in diseases, solving the human PAF-R structure, and identifying PAF signalling mechanisms independent of the PAF-R are some of the many major achievements in current PAF research.